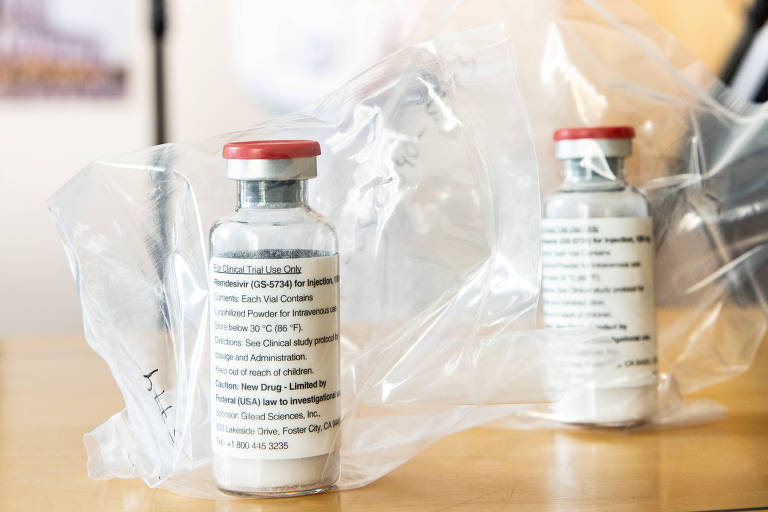
Gilead, the American laboratory manufacturer of the antiviral remdesivir, shown to produce moderately positive results in the treatment against Covid-19, has waived the patent on the drug to facilitate its access in 127 countries. Brazil, however, was excluded from the list.
In a statement released on Tuesday (12), the company said it had signed voluntary licensing agreements with five pharmaceutical companies specializing in the production of generics, all based in India or Pakistan.
"Under the licensing agreement, companies are entitled to receive a technology transfer from remdesivir's manufacturing process so that production can scale more quickly," Gilead said in the statement.
By giving up the patent, Gilead will help to make the drug more readily available and cheaper across the world.
The companies that signed the agreement with the laboratory are Cipla, Ferozsons, Hetero, Jubilant, and Mylan. They will be able to fix the price of the generic product for sale in the countries where they operate, most of them impoverished nations in Latin America, Asia, and Africa.
The list also includes some middle-income countries, regional powers, and emerging states, such as South Africa, Egypt, Nigeria, India, Indonesia, Pakistan, Thailand, and Ukraine.
Rich countries were left out. In South America, only Guyana and Suriname were included.
The laboratory told Folha that it based its decision on countries covered by voluntary licensing on the World Bank's list that defines low and middle-income countries, "with the inclusion of some exceptions."
There was no specific explanation of why Brazil was left out.
Translated by Kiratiana Freelon