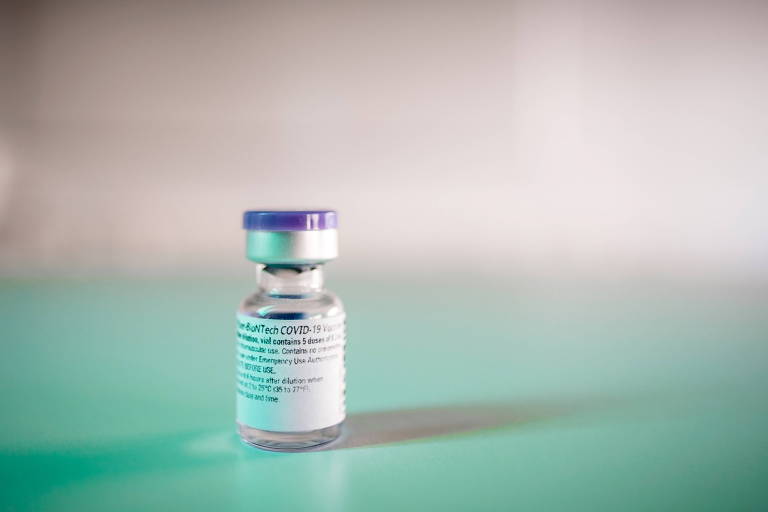
The Ministry of Health reported on Tuesday (1) that vaccines against Covid-19 that will be included in the National Immunization Plan must "fundamentally" be thermostable and be able to be stored at temperatures from 2 ° C to 8 ° C.
In practice, the announcement means that the vaccine developed by the American Pfizer and the German BioNTech will not be applicable in Brazil since it requires special storage conditions, with temperatures of -70º C.
On Tuesday, Pfizer asked European drug regulatory authorities for authorization for the emergency use of its vaccine. The same process had already started in the United States. Pfizer has released studies showing that its vaccine is 95% effective against the new coronavirus.
In a press conference, the Ministry's Health Surveillance Secretary, Arnaldo Medeiros, stated that it is desirable that the vaccine be applied in a single dose and that it is also "fundamentally" thermostable for long periods, at temperatures of 2 ° C to 8 ° C. The representative of the portfolio did not specifically mention any vaccine.
Translated by Kiratiana Freelon